UNIT 6: Drug Challenge and Clinical Evaluation Concepts
This unit explains how drug challenge information is used in Pharmacovigilance to clinically evaluate the relationship between a medicinal product and an adverse event. It focuses on dechallenge and rechallenge concepts, their outcomes, limitations, and real-world regulatory interpretation rather than theoretical assumptions.
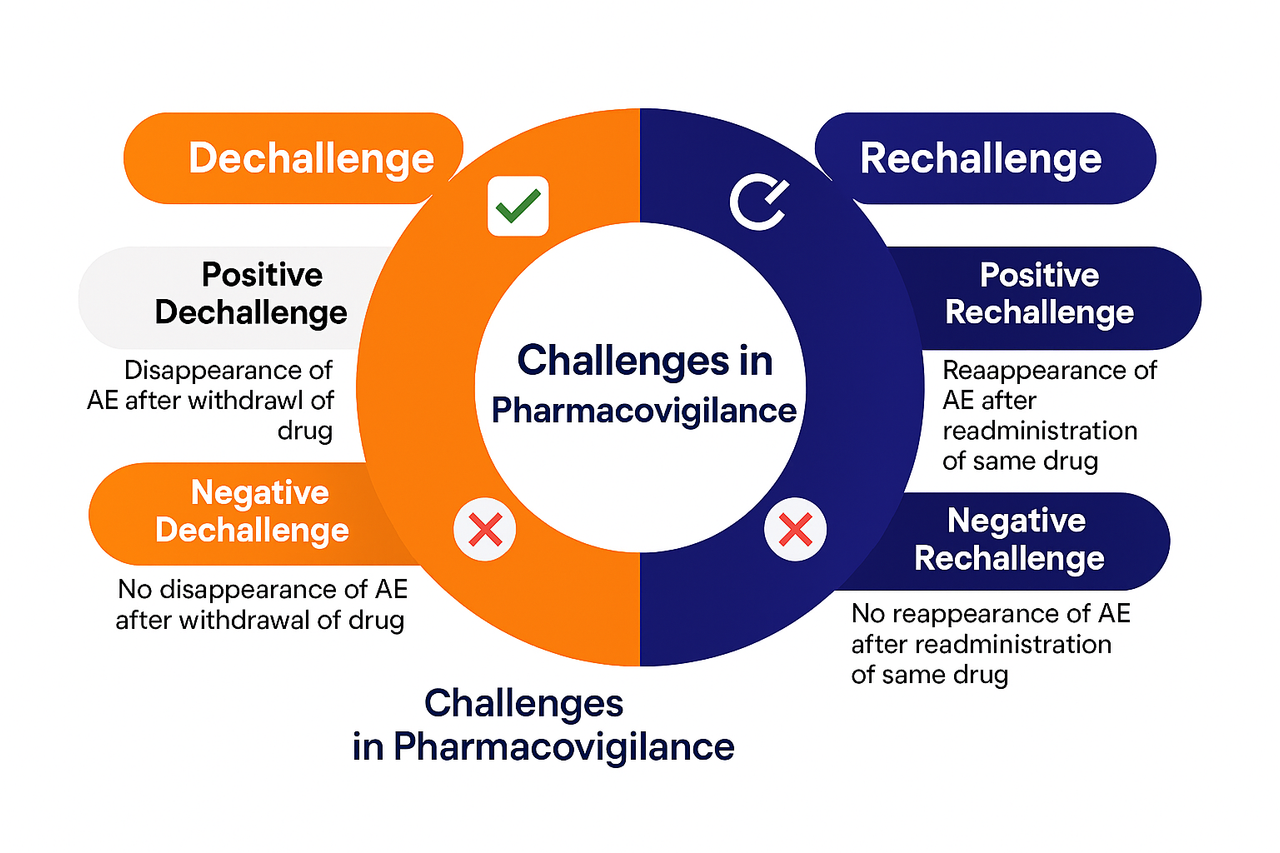
Drug challenge information plays an important supportive role in pharmacovigilance by helping to evaluate whether a medicinal product is related to an adverse event. It is not a standalone proof of causality but is used together with clinical history, temporal relationship, known pharmacology, and alternative explanations.
Key Points
Drug challenge supports, but does not confirm causality alone.
Used mainly in clinical evaluation of cases.
Must always be interpreted with caution.
How was this unit?
Your feedback helps us improve our content