UNIT 4: CTD and eCTD Structure for Regulatory Submissions
This unit provides a structured understanding of the Common Technical Document (CTD) and electronic Common Technical Document (eCTD) frameworks used globally for regulatory submissions. It explains the purpose and content of each CTD module with clear linkage to regulatory expectations.
Definition
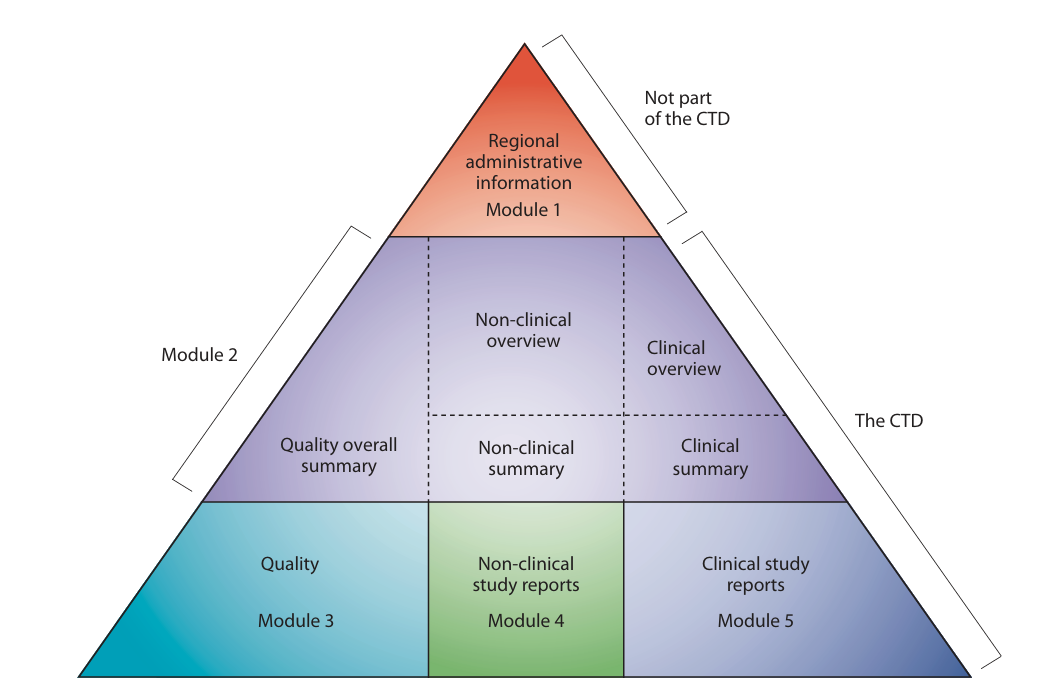
The Common Technical Document (CTD) is a harmonized format developed by the International Council for Harmonisation to organize quality, nonclinical, and clinical data for regulatory submissions. The electronic CTD (eCTD) is the electronic version of the CTD used for lifecycle management of submissions.
CTD defines what content is submitted, while eCTD defines how the content is organized, structured, and maintained electronically throughout the product lifecycle. Most global regulatory authorities now mandate eCTD for submissions.
| Region | Submission Format Requirement |
|---|---|
| United States | eCTD mandatory |
| European Union | eCTD mandatory |
| United Kingdom | eCTD mandatory |
| Japan | eCTD mandatory |
| Canada | eCTD mandatory |
| India | CTD / eCTD (transition phase) |
Explore Our Courses
Master life sciences with comprehensive courses in regulatory science, clinical research, and pharmacovigilance.
How was this unit?
Your feedback helps us improve our content